FRONTIERS IN MEDICAL CASE REPORTS - Volume 7; Issue 1, (Jan-Feb, 2026)
Pages: 01-10
Print Article
Download XML Download PDF
Spectral EEG-Guided Adaptive Neuromodulation for Age-Related Cognitive Changes: A Longitudinal Case Report
Author: Yatharth Mahajan, Mark Odron, Vipul Reddy, Krrishika Saxena, Netra Ramanatham, Brianna Dela Cruz, Athreykrishna Parla, Charles Vigilia, Kenneth Blum, Rajendra D. Badgaiyan, Keerthy Sunder
Category: Clinical Case Reports
Abstract:
Targeted interventions for age-related cognitive changes in older adults are complicated by co-occurring factors such as depression, anxiety, executive dysfunction, and treatment heterogeneity, necessitating strategies that integrate cognitive, affective, and functional symptom management. This case report explores functional, affective, sleep, and quality of life outcomes following EEG-spectra-guided personalized repetitive transcranial magnetic stimulation (PrTMS, PeakLogic, Inc., San Diego, CA) for a patient experiencing age-related cognitive changes. Weekly assessments showed improvements in perceived cognitive function, sleep, mood, and quality of life, quantified via Perceived Deficits Questionnaire - Depression (PDQ-D5), Sleep Condition Indicator (SCI), Patient Health Questionnaire-9 (PHQ-9), Generalized Anxiety Disorder 7-Item Scale (GAD-7), and Quality of Life Enjoyment and Satisfaction Questionnaire Short Form (Q-LES-Q-SF). Our findings show promise that EEG-spectra-guided PrTMS is potentially beneficial for addressing age-related cognitive and functional changes. Furthermore, our findings show longitudinal symptom improvements consistent with engagement of adaptive neuroplasticity. Further large-scale, randomized and blinded studies are recommended to validate our observations on the feasibility of PrTMS for age-related cognitive changes.
Keywords: Personalized Repetitive Transcranial Magnetic Stimulation (PrTMS), Dorsolateral Prefrontal Cortex (DLPFC), Neuromodulation, Normal Cognitive Aging, Perceived Deficits Questionnaire - Depression (PDQ-D5), Spectral Electroencephalography (EEG)
DOI URL: https://dx.doi.org/10.47746/FMCR.2026.7103
Full Text:
Introduction
The study focuses on personalized repetitive transcranial magnetic stimulation (PrTMS) as a modality to improve age-related cognitive and functional changes in a healthy elderly patient. Normal cognitive aging is characterized by gradual decline in fluid intelligence and relatively preserved crystallized intelligence, which may present as memory lapses, misplacing objects, and difficulty with problem solving (Murman, 2015). Additionally, this process involves a flattened diurnal rhythm of cortisol release and reduced melatonin production, which decreases total sleep time and contributes to hyperarousal (Pandi-Perumal et al., 2005; Yiallouris et al., 2019). These findings are associated with changes in neurotransmitter pathways, particularly involving dopamine which plays a key role in cognitive processes such as memory, attention, and reasoning (Lee and Kim, 2022). Such changes in older adults are generally mild and slowly progressive, and do not interfere with the ability to accomplish daily tasks, distinct from pathologic conditions such as mild cognitive impairment (MCI) and dementia (Moreira et al., 2019; Saxena and Nettles, 2022; Yan et al., 2023).
Repetitive transcranial magnetic stimulation (rTMS) is a non-invasive intervention that modulates cortical excitability and promotes neuroplasticity, with evidence suggesting that high-frequency stimulation of the dorsolateral prefrontal cortex (DLPFC) can improve working memory, attention, and executive function in older adults (Miniussi et al., 2013), and a more recent study suggesting that it may even promote reversal of MCI to normal cognition (Guo et al., 2025). In the context of dementia, rTMS has shown clinically meaningful benefits. A meta-analysis of 14 trials involving 655 elderly patients with either MCI or dementia revealed modest but significant improvements in global cognitive performance in rTMS groups compared to sham groups, quantified via Mini-Mental State Examination (MMSE), Montreal Cognitive Assessment (MoCA), and Alzheimer's Disease Cognitive Subscale (ADAS-Cog) (Cao and Yang, 2025). These results support rTMS as a safe and potentially feasible intervention for cognitive deficits, while highlighting the need for further trials to optimize treatment protocols.
PrTMS is a novel form of neuromodulation involving low-intensity stimulation across multiple scalp sites, rather than high-intensity stimulation to a single site (e.g. left DLPFC) seen in many standard rTMS protocols. To address individual variability in neurophysiology that may limit standard rTMS protocols, PrTMS integrates patient-specific parameters such as weekly spectral electroencephalograms (EEGs) and symptom-based questionnaires to individualize stimulation parameters (Makale et al., 2024). It has shown promising results in patients with depression, anxiety, PTSD, insomnia, concussion, and autism spectrum disorder (ASD) (Mohankumar et al., 2025). Although validated symptom-based neuropsychiatric questionnaires are commonly used to assess cognitive function in the elderly, they are rarely incorporated into adaptive neuromodulation therapies. Most rTMS protocols apply fixed stimulation parameters without accounting for longitudinal symptom variability, which may limit treatment optimization in normal older adults. This represents a gap in neuromodulation applications that the study aims to explore.
Case History
Patient is a 73-year old African American female who reported periods of slower thought processes, difficulty multitasking, occasional word-finding deficits, poor focus, and minor memory lapses, which she stated had caused difficulty in accomplishing occupational tasks as a medical receptionist. She recalled onset of symptoms approximately 10 years ago, along with more recent onset of quality of life impairments due to poor sleep, limited mobility, and interpersonal conflicts. She also reported recent onset of depressive symptoms such as low mood, anhedonia, guilt, and psychomotor slowing, which she attributed to these limitations. Despite these symptoms, the patient reported adequate ability to accomplish basic and instrumental activities of daily living (ADLs) independently. Cognitive screening was unremarkable, with MMSE and MoCA showing deficits in serial 7s, but scoring 29 and 28 / 30, respectively. This suggests largely preserved global cognition and a low likelihood of MCI and dementia. PrTMS was recommended in mid-2025 to alleviate her symptoms and improve daily function and quality of life.
Methods
Subject
The subject was selected based on age ≥65 years and no evidence of MCI or dementia. She had no contraindications to TMS, such as ferromagnetic implants within 12 inches of the head, cardiac pacemaker, implantable cardioverter defibrillator, and history of epilepsy or brain lesions (Mann and Malhi, 2025). The subject was recommended 50 once-daily PrTMS sessions over 3 months. She did not take any neuropsychiatric medications or do cognitive training for the duration of the study.
Personalized Repetitive Transcranial Magnetic Stimulation (PrTMS) Treatment
PrTMS was administered by certified neurotechnologists using the PeakLogic software installed in the Apollo TMS Therapy System. Treatment locations include Cz (central midline), Fz (frontal midline), F3 (left dorsolateral prefrontal cortex), F4 (right dorsolateral prefrontal cortex), and Fpz (prefrontal midline). The total treatment time was approximately 30 minutes per session. Stimulation parameters, including amplitude and frequency, were adjusted on a weekly basis via computerized algorithm (PeakInput), which generates treatment protocols based on weekly EEG measurements and symptom-based questionnaires (Makale et al., 2024).
Spectral EEG Measurements
Spectral EEG was performed on a weekly basis using a CGX high-impedance dry electrode headset to monitor changes in brain wave activity over time. Electrodes were positioned according to the 10-20 system. It was also performed after a 2-month naturalistic follow-up period.
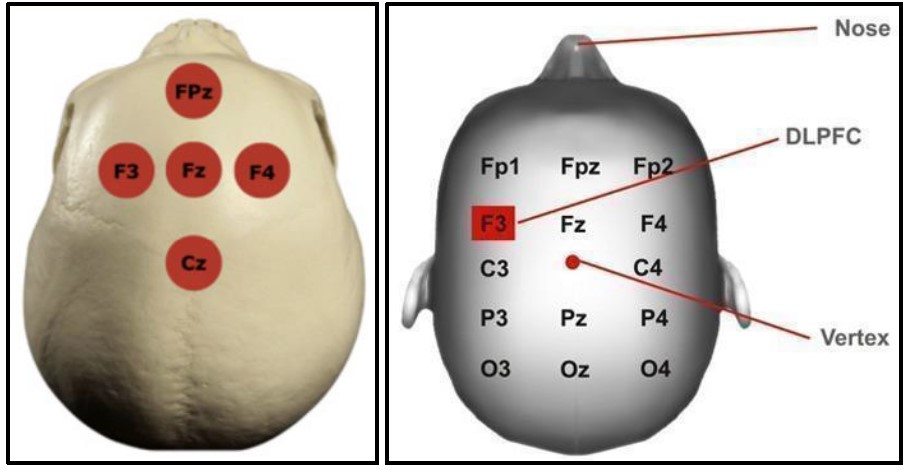
Figure 1: PrTMS locations. Figure 2: 10-20 system locations.
Psychometric and Cognitive Questionnaire Administration
Symptoms were monitored using standardized psychometric and cognitive questionnaires. The Perceived Deficits Questionnaire - Depression (PDQ-D5) was administered as a reliable measure to capture subjective cognitive complaints in depressed patients (Sumiyoshi et al., 2022). Additionally, the Patient Health Questionnaire-9 (PHQ-9) and Generalized Anxiety Disorder 7-Item Scale (GAD-7) were administered as reliable psychometric tools for monitoring depressive symptoms and anxiety (Pranckeviciene et al., 2022). The Sleep Condition Indicator (SCI) was administered as a validated screening tool to assess frequency and severity of insomnia (Meng et al., 2025). The Quality of Life Enjoyment and Satisfaction Questionnaire Short Form (Q-LES-Q-SF) was administered as a validated measure of health-related quality of life and overall satisfaction (Rush et al., 2019). The Six Item Cognitive Impairment Test (6CIT) was also administered to periodically assess the need for further screening for MCI and dementia (Abdel-Aziz and Larner, 2015). These surveys were given on a weekly basis over a 3-month period between 8/19 and 11/12/2025, and after a 2-month naturalistic follow-up period (1/13/2026).
Results
Table 1: Weekly scores for psychometric and cognitive questionnaires.
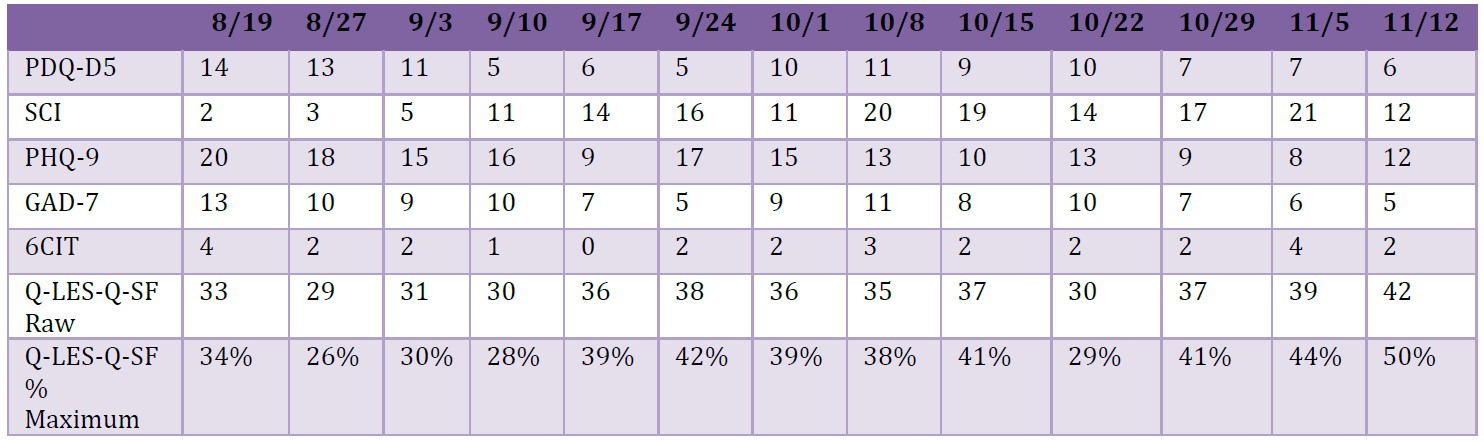
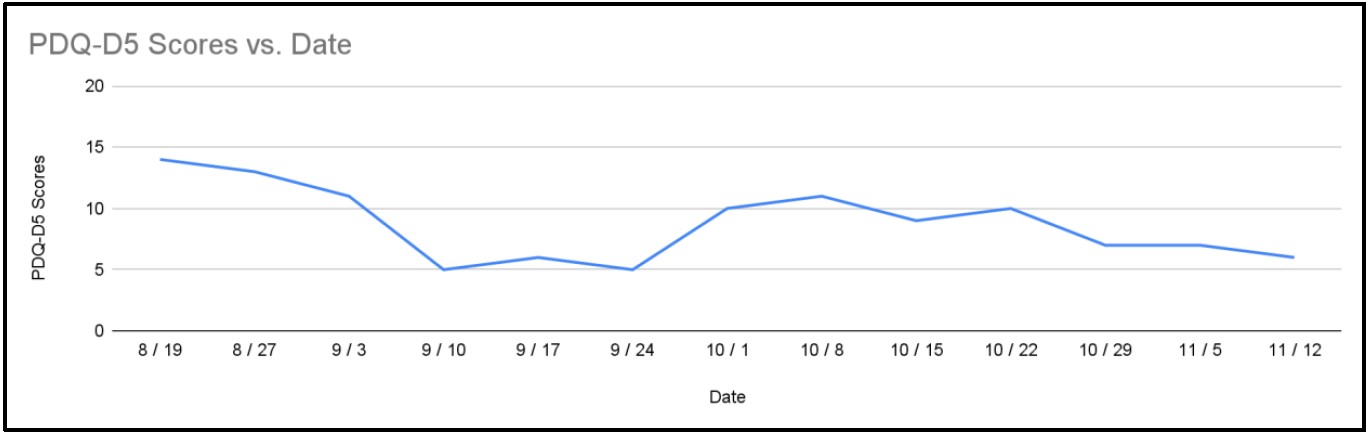
Figure 3: Weekly PDQ-D5 scores.
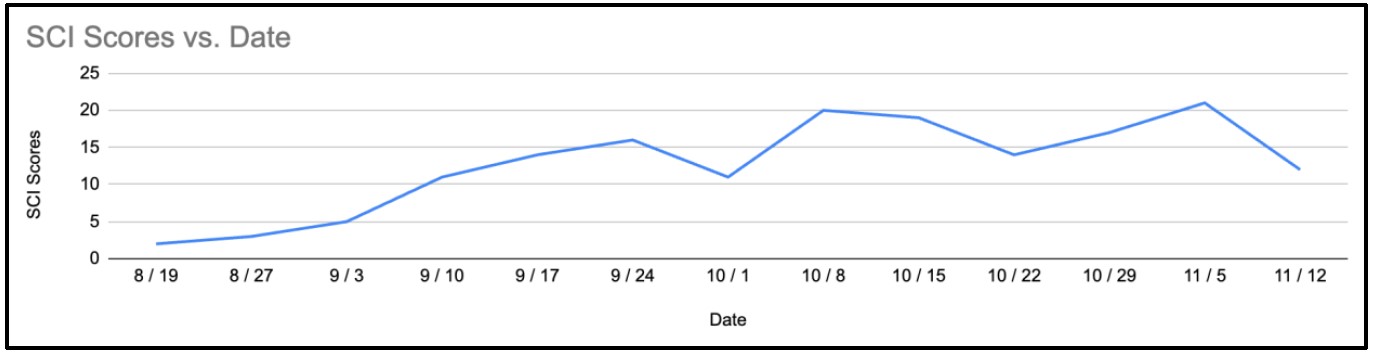
Figure 4: Weekly SCI scores.
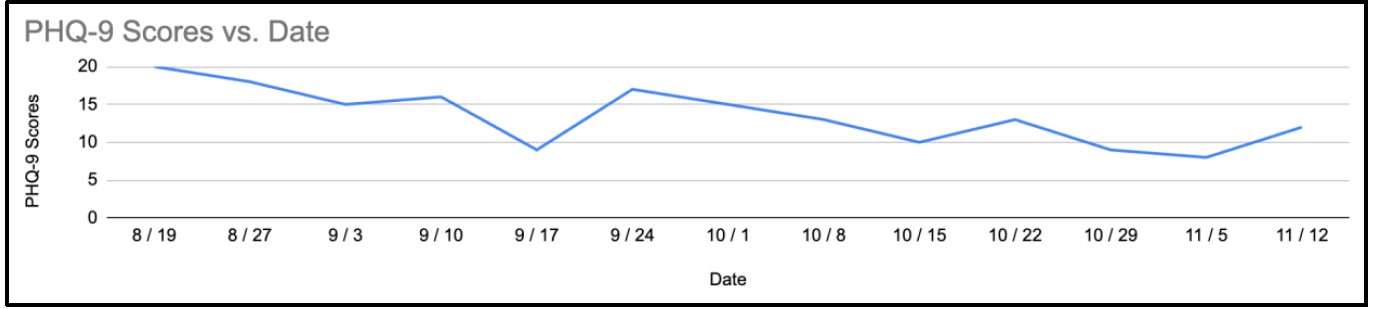
Figure 5: Weekly PHQ-9 scores.
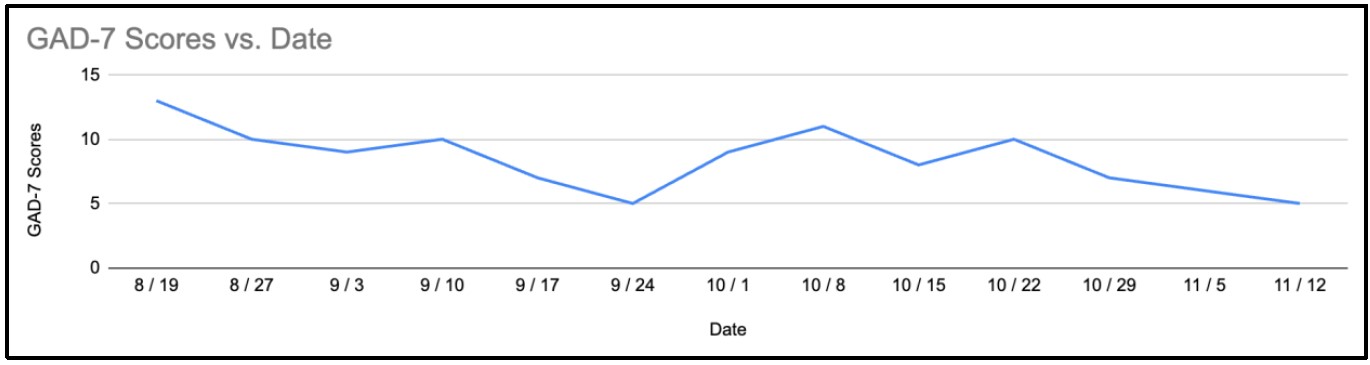
Figure 6: Weekly GAD-7 scores.
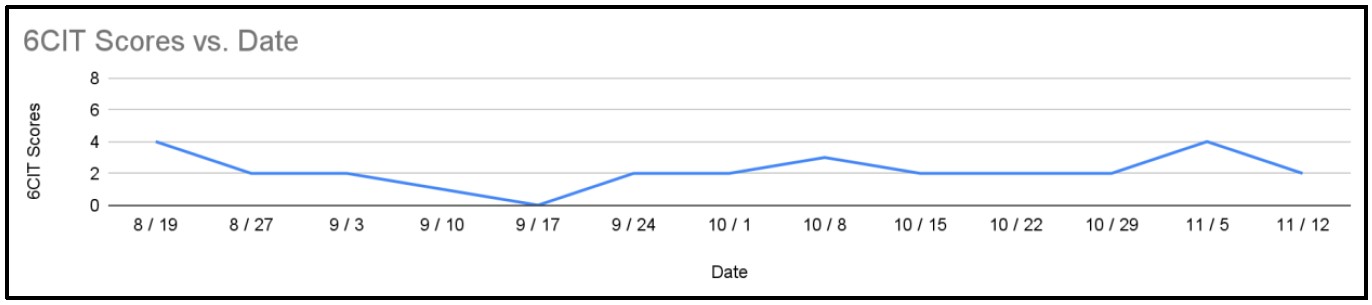
Figure 7: Weekly 6CIT scores.
Table 2: Psychometric and cognitive questionnaire scores after the 2-month follow-up period.

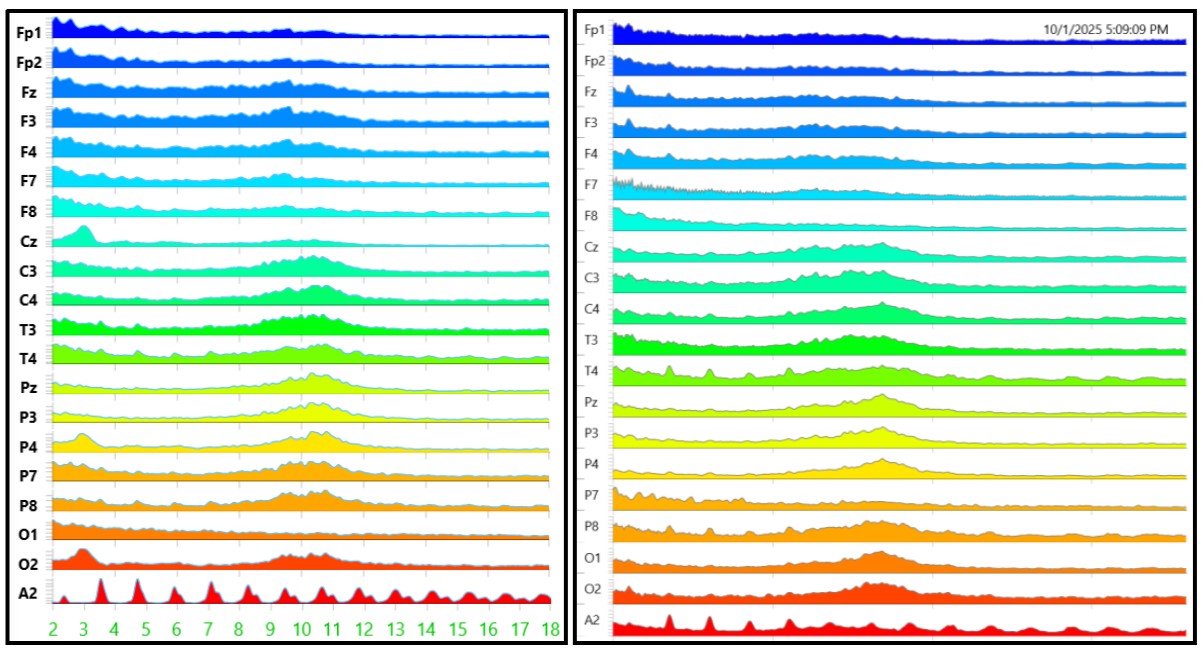
Figure 8: Initial EEG (8/18/2025). Figure 9: Midpoint EEG (10/1/2025).
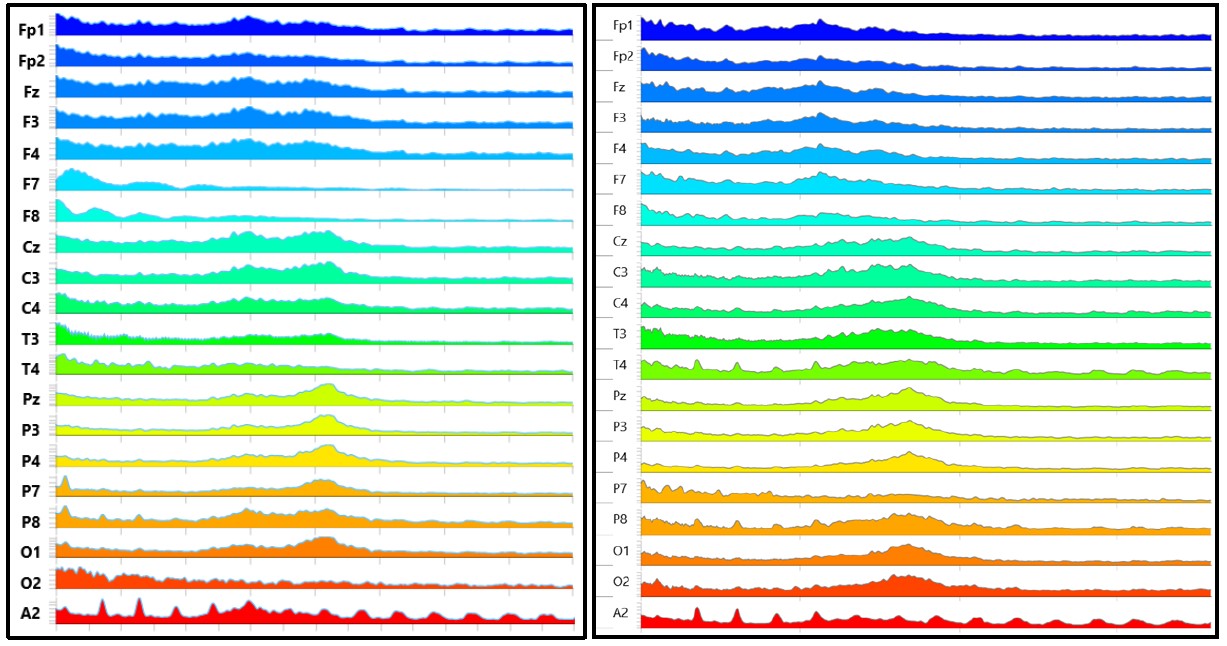
Figure 10: EEG on final day of treatment. Figure 11: Follow-up EEG (1/28/2026).
Discussion
During the PrTMS treatment course and follow-up period, substantial and consistent improvements across cognitive, affective, and functional domains were observed. Cohen’s d was utilized, revealing within-patient effect sizes that exceeded the threshold for large clinical impact (d > 0.8) (Becker 2000). The objectives of the interventions were to reduce age-related cognitive changes and improve depressive symptoms, anxiety, and sleep quality. Large standardized reductions were observed for the PDQ-D5 (d = -2.23), PHQ-9 (d = -2.03), and GAD-7 (d = -2.30). These values coincide with self-reported decreases in perceived cognitive lapses, depressive symptoms, and anxiety. Conversely, substantial improvements were observed in measures where higher scores indicate functional gains, specifically the SCI (d = +2.14) and Q-LES-Q-SF (+0.52). The use of these psychometrics in conjunction with spectral EEGs on a weekly basis allowed for dynamic adjustment of stimulation parameters based on individual symptomatology. This ensured that low-amplitude, multi-site stimulation remained synchronized with the patient's evolving cortical state (Makale et al., 2023). The patient reported improvements in cognitive processing, fluid memory, sleep consolidation, and depressive symptoms, which may be associated with adaptive neuroplastic changes and alterations in cortical excitability. She also reported modest improvements in satisfaction across multiple quality of life domains, including daily and occupational functioning, family and social relationships, and future outlook, consistent with improvements in weekly Q-LES-Q-SF scores.
Additionally, changes in alpha wave amplitude were noted over the treatment course. Alpha waves range in frequency from 8 to 12 Hz. Alpha oscillations, particularly in frontal leads (Fz, F3, F4, Fp1, and Fp2), are commonly interpreted as indices of cortical efficiency, calm alertness, creativity, focus, and top-down control (Lustenberger et al., 2015; Mahajan et al., 2025). Age-related decreases in alpha wave amplitude have been associated with diminished selective attention and executive function (Lejko et al., 2020). Low alpha band activity in the prefrontal cortex has also been associated with increased sleep fragmentation and poorer subjective sleep quality in adults (Han et al., 2024). The patient’s subjective symptom improvements were accompanied by slow but progressive increases in frontal alpha power, determined qualitatively via serial spectral EEG readings over the 12-week treatment course and follow-up period.
Limitations
The use of PrTMS for age-related cognitive changes was limited to a single case study. Its feasibility must be further studied using large-scale, randomized and blinded trials in order to validate findings. Another limitation is the use of self-reported psychometric questionnaires, which are inherently subject to response bias and placebo effects. To contextualize these subjective outcomes, serial longitudinal spectral EEGs were administered as a more objective measure of neurophysiologic activity.
Conclusion
This study highlights a case wherein individualized neuromodulation via PrTMS was followed by clinically meaningful and sustained improvements in an elderly patient experiencing normal cognitive aging. Quantitative analysis revealed substantial improvements in psychometric scores, specifically the PDQ-D5 (d = -2.23), PHQ-9 (d = -2.03), GAD-7 (d = -2.30), SCI (d = +2.14) and Q-LES-Q-SF (d = +0.52). These results suggest that tailoring stimulation to individualized neurophysiologic profiles via weekly spectral EEG monitoring may potentially address the heterogeneous patterns of cortical network inefficiency. Compared to standard left DLPFC stimulation, this adaptive approach may offer a more optimized method for modulating brain wave activity, potentially resulting in more robust long-term improvement and durability. Further large-scale, blinded, and randomized studies are recommended to better understand the role of PrTMS as a potential intervention to optimize outcomes for normal cognitive aging.
Author Contributions
K. Sunder conceptualized the study. The original draft of this manuscript was written by YM, MO, and K. Sunder. Review and additional editing of the manuscript were conducted by VR, K. Saxena, NR, BDC, AP, CV, KB, and RB.
Acknowledgements: KB is the recipient of R41 MD012318 / MD / NIMHD NIH HHS / United States, and I01 CX000479 / CX / CSRD VA / United States. The funding sources had no role in the design, data collection, analysis, or preparation of this manuscript.
Conflict of Interest: We report no potential conflicts of interest regarding the publication of this paper. This manuscript has been read and approved by all authors.
Declaration of Patient Consent: The authors confirm that written informed consent was obtained from the participant for publication of this paper. Participation in the study adhered strictly to patient privacy and HIPAA guidelines. The participant understood the potential risks and benefits of the interventions.
References:
Abdel-Aziz K, Larner AJ. Six-item cognitive impairment test (6CIT): pragmatic diagnostic accuracy study for dementia and MCI. Int Psychogeriatr 2015; 27: 991-997.
Becker Lee A. "Effect size (ES)." (2000): 2000.
Cao ZG, Yang M. Repetitive Transcranial Magnetic Stimulation for Elderly Patients with Cognitive Impairment: A Meta Analysis of Randomized Controlled Trials. J Neurol Neuromedicine 2025; 9: 1-21
Guo Z, Jiang Y, He J, Jiang N. Repetitive transcranial magnetic stimulation may promote the reversion of mild cognitive impairment to normal cognition. Front Psychiatry 2025; 16: 1544728.
Han C, Zhang Z, Lin Y, Huang S, Mao J, Xiang W, Wang F, Liang Y, Chen W, Zhao X. Monitoring Sleep Quality Through Low α-Band Activity in the Prefrontal Cortex Using a Portable Electroencephalogram Device: Longitudinal Study. J Med Internet Res 2025; 27: e67188.
Lee J, Kim HJ. Normal Aging Induces Changes in the Brain and Neurodegeneration Progress: Review of the Structural, Biochemical, Metabolic, Cellular, and Molecular Changes. Front Aging Neurosci 2022; 14: 931536.
Lejko N, Larabi DI, Herrmann CS, Aleman A, Ćurčić-Blake B. Alpha Power and Functional Connectivity in Cognitive Decline: A Systematic Review and Meta-Analysis. J Alzheimers Dis 2020; 78: 1047-1088.
Lustenberger C, Boyle MR, Foulser AA, Mellin JM, Fröhlich F. Functional role of frontal alpha oscillations in creativity. Cortex 2015; 67: 74-82.
Mahajan S, Bodahanapati A, Katta A, Aydemir E, Mendoza A, Vigilia C, Blum K, Baron D, Lewandrowski KU, Badgaiyan RD, Sunder K. Personalized Repetitive Transcranial Magnetic Stimulation (PrTMS®) Coupled with Transcranial Photobiomodulation (tPBM) For Co-Occurring Traumatic Brain Injury (TBI) and Post-Traumatic Stress Disorder (PTSD). Acta Sci Neurol 2025; 8: 20-27.
Makale MT, Nybo C, Blum K, Dennen CA, Elman I, Murphy KT. Pilot Study of Personalized Transcranial Magnetic Stimulation with Spectral Electroencephalogram Analyses for Assessing and Treating Persons with Autism. J Pers Med 2024; 14: 857.
Mann SK, Malhi NK. Repetitive Transcranial Magnetic Stimulation. 2023 Mar 6. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025. PMID: 33760474.
Meng R, Huang M, Miller CB, Fong DYT, Gregory AM, Pakpour AH, Dzierzewski JM, Henry AL, Voinescu BI, Yang N, Ma H, Luo Y, Lau EYY, Spruyt K, Espie CA. Global perspectives on the Sleep Condition Indicator for DSM-5 insomnia disorder: a COSMIN and STARD systematic review of psychometric and diagnostic performance. BMC Med 2025; 23: 542.
Miniussi C, Harris JA, Ruzzoli M. Modelling non-invasive brain stimulation in cognitive neuroscience. Neurosci Biobehav Rev 2013; 37: 1702-1712.
Mohankumar K, Karthikeya S, Mahajan S, Bodhanapati J, Vigilia C, Odron M, Blum K, Baron D, Lewandrowski KU, Badgaiyan RD, Modestino EJ, Sunder K. EEG-Spectra-Guided Personalized rTMS in PTSD with Co-occurring Psychiatric Disorders: A Case Series. Acta Sci Neurol 2025; 8: 3-9.
Moreira HS, Costa AS, Machado Á, Castro SL, Lima CF, Vicente SG. Distinguishing mild cognitive impairment from healthy aging and Alzheimer's Disease: The contribution of the INECO Frontal Screening (IFS). PLoS One 2019; 14: e0221873.
Murman DL. The Impact of Age on Cognition. Semin Hear 2015; 36: 111-121.
Pandi-Perumal SR, Zisapel N, Srinivasan V, Cardinali DP. Melatonin and sleep in aging population. Exp Gerontol 2005; 40: 911-925.
Pranckeviciene A, Saudargiene A, Gecaite-Stonciene J, Liaugaudaite V, Griskova-Bulanova I, Simkute D, Naginiene R, Dainauskas LL, Ceidaite G, Burkauskas J. Validation of the patient health questionnaire-9 and the generalized anxiety disorder-7 in Lithuanian student sample. PLoS One 2022; 17: e0263027.
Rush AJ, South CC, Jha MK, Grannemann BD, Trivedi MH. Toward a very brief quality of life enjoyment and Satisfaction Questionnaire. J Affect Disord 2019; 242: 87-95.
Saxena K, Nettles S (2022). Understanding Alzheimer’s risk factors associated with the prevalence of US populations and women. Journal of Student Research 2022; 11(1).
Sumiyoshi T, Uchida H, Watanabe K, Oosawa M, Ren H, Moriguchi Y, Fujikawa K, Fernandez J. Validation and Functional Relevance of the Short Form of the Perceived Deficits Questionnaire for Depression for Japanese Patients with Major Depressive Disorder. Neuropsychiatr Dis Treat 2022; 18: 2507-2517.
Yan Y, Tian M, Wang T, Wang X, Wang Y, Shi J. Transcranial magnetic stimulation effects on cognitive enhancement in mild cognitive impairment and Alzheimer's disease: a systematic review and meta-analysis. Front Neurol 2023; 14: 1209205.
Yiallouris A, Tsioutis C, Agapidaki E, Zafeiri M, Agouridis AP, Ntourakis D, Johnson EO. Adrenal Aging and Its Implications on Stress Responsiveness in Humans. Front Endocrinol (Lausanne) 2019; 10: 54.
|










