FRONTIERS IN MEDICAL CASE REPORTS - Volume 7; Issue 2, (Mar-Apr, 2026)
Pages: 01-07
Print Article
Download XML Download PDF
Lamotrigine-Induced Stevens-Johnson Syndrome in a Young Female Patient with Atypical Anorexia Nervosa and Major Depression: A Case Report
Author: Miguel Almeida-Pérez, Elizabeth Valdivia-Marquez, Karina García-Pérez, Deysi Carhuamaca-Lino, David Gómez-Pereda
Category: Medical Case Reports
Abstract:
Stevens-Johnson Syndrome (SJS) is a rare, severe cutaneous adverse reaction often triggered by medications such as lamotrigine. We present the case of a 21-year-old female with Atypical Anorexia Nervosa and Major Depression who developed SJS thirty days after initiating a psychopharmacological regimen comprising lamotrigine, fluoxetine, and olanzapine. Clinical examination revealed extensive mucocutaneous lesions, including hemorrhagic cheilitis, palpebral ulcers, and targetoid lesions. The condition resolved following immediate drug discontinuation and systemic corticosteroid treatment. This case underscores the risks of rapid titration and polypharmacy in patients with Atypical Anorexia Nervosa, where metabolic instability may alter drug metabolism. Clinicians must exercise heightened vigilance and strictly adhere to slow titration schedules in this vulnerable population.
Keywords: Stevens-Johnson Syndrome, Lamotrigine, Anorexia Nervosa, Major Depression, Polypharmacy, Drug Hypersensitivity
DOI URL: https://dx.doi.org/10.47746/FMCR.2026.7201
Full Text:
Introduction
Stevens-Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TEN) represent a spectrum of severe cutaneous adverse reactions (SCARs) characterized by widespread epidermal necrosis and mucosal detachment (Harr and French, 2010; Schwartz et al., 2013; Stern, 2014). The pathophysiology involves a T-cell-mediated type IV hypersensitivity reaction (Chung and Hung, 2012).
Among the various drug classes implicated, lamotrigine carries a "black box" warning for serious rashes. The risk of lamotrigine-induced SJS is approximately 0.08% to 0.13% in adults, with a higher incidence observed during rapid titration (Edinoff et al., 2021; Mockenhaupt et al., 2005; Roujeau et al., 1995). Despite these warnings, lamotrigine use in psychiatry continues to rise. Clinical practice often involves patients with complex comorbidities like Atypical Anorexia Nervosa, which induces significant metabolic stress even in the absence of low body weight (Winston, 2012).
Currently, there is a gap in the literature regarding how the metabolic instability of atypical eating disorders might influence susceptibility to SCARs. This case report addresses this gap by describing a life-threatening presentation of SJS in a young female with Atypical Anorexia Nervosa, highlighting the mechanistic interactions between erratic nutrition, preventable prescribing errors in polypharmacy, and drug hypersensitivity.
Case Description
Clinical History and Presentation
A 21-year-old female presented to the Emergency Department following a four-day prodromal phase of fever, malaise, and a stinging sensation in the eyes, progressing to painful oral erosions. Her medical history was notable for Atypical Anorexia Nervosa (diagnosed 6 years prior; pre-admission BMI 21.5 kg/m², without a history of an extreme low-weight nadir), Recurrent Major Depressive Disorder, and chronic gastritis. She had no prior drug allergies.
Due to severe acute depressive decompensation, her psychiatric regimen was aggressively adjusted thirty days prior to admission. No sertraline was administered at any point. Her drug regimen at symptom onset (Day 0) was as follows:
• Lamotrigine: Initiated Day -30 (25 mg/day). Escalated Day -19 (100 mg/day). Escalated Day -14 (125 mg/day). Escalated Day -12 (150 mg/day). Discontinued Day 0.
• Fluoxetine: 40 mg/day. Initiated 6 months prior. Discontinued Day 0.
• Olanzapine: 5 mg/night. Initiated Day -30. Discontinued Day 0.
Physical Examination
Upon admission, the patient was tachycardic (110 bpm) and febrile (37.8°C).
• Mucocutaneous: Extensive hemorrhagic crusting of the lips and buccal erosions.
• Ocular: Unilateral right palpebral ulcer and conjunctivitis.
• Dermatological: Erythematous, targetoid maculopapular lesions on the palms and soles. The Nikolsky sign was negative.
• Genitourinary: Painful ulcerative lesions on the vulvar mucosa.
Given the classic, unambiguous clinical presentation and to minimize patient discomfort, a skin biopsy was not performed.
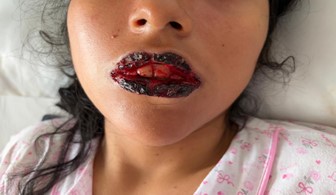
Figure 1: Clinical photograph showing extensive hemorrhagic cheilitis and oral mucosal erosion at admission.
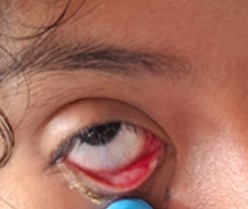
Figure 2: Image displaying conjunctivitis with ulcer and serohematic discharge.
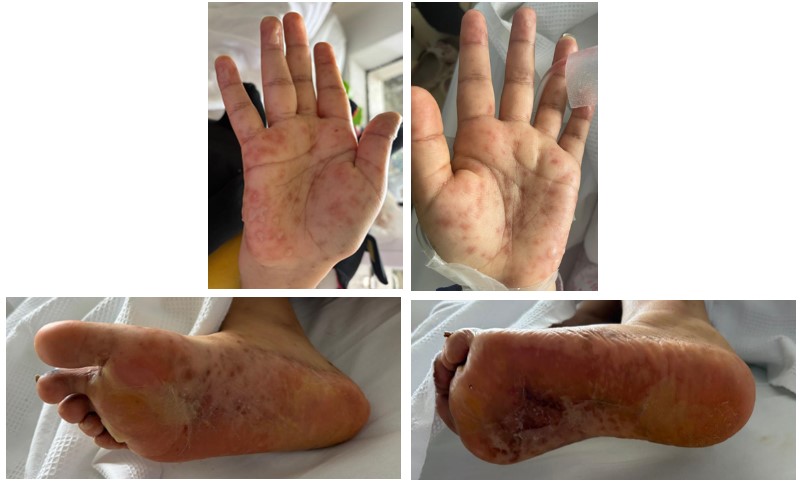
Figure 3: Photograph of the patient's palms The plantar surface exhibits diffuse erythema, hyperkeratosis, and desquamation with scattered dark brown macules.
Diagnostic Assessment
Diagnosis of SJS was made clinically based on the timeline and characteristic mucocutaneous detachment (Schwartz et al., 2013). Laboratory investigations revealed mild leukocytosis (12,400/mm³). Serum albumin remained at the lower limit of normal (3.47 g/dL) throughout her admission, reflecting baseline metabolic stress from erratic dietary patterns. Specific oxidative stress or glutathione markers were unavailable in our acute setting.
The Naranjo Scale indicated a "probable" adverse drug reaction to lamotrigine (Score = 6: +1 previous conclusive reports, +2 temporal sequence, +1 improvement upon discontinuation, +2 alternative causes excluded) (Naranjo et al., 1981). Additionally, the ALDEN (algorithm of drug causality for epidermal necrolysis) score was 6, indicating a "highly probable" causality for lamotrigine.
Therapeutic Intervention and Outcomes
Following the cessation of all psychotropic medications, she received systemic corticosteroids (Dexamethasone 4 mg IV every 8 hours for 3 days), aggressive hydration, and local mucosal care. Cutaneous progression halted within 72 hours, and she was discharged on day 14 with almost complete resolution.
Discussion
Diagnostic Criteria and HLA Associations
SJS pathogenesis involves a specific immune reaction where the drug binds to human leukocyte antigen (HLA) molecules (Chung and Hung, 2012). While alleles like HLA-B*1502 are genetic risk factors, SJS diagnosis remains primarily clinical and does not require HLA testing (Chung and Hung, 2012; McCormack et al., 2011). The classic timeline in this case-symptoms appearing four weeks after drug initiation—was sufficient for diagnosis.
Titration Protocols, Preventable Errors, and Interactions
Standard guidelines mandate that lamotrigine must be titrated very slowly-starting at 25 mg daily for two weeks, followed by 50 mg daily for the next two weeks (Mockenhaupt et al., 2005). The abrupt quadrupling of this patient’s dose to 100 mg on day 11, while reportedly driven by severe depressive urgency, represents a preventable prescribing error that directly violated safety protocols and triggered the SCAR. Furthermore, concurrent polypharmacy compounded this risk; fluoxetine is a known CYP450 inhibitor, and its concurrent use alongside olanzapine may have competitively inhibited lamotrigine clearance, elevating its plasma levels (Spina and de Leon, 2007).
The Role of Atypical Anorexia Nervosa
Atypical Anorexia Nervosa presents unique pharmacological challenges (Winston, 2012). Despite a normal BMI, erratic dietary intake can lead to transient micronutrient deficiencies, including the depletion of antioxidants like glutathione. While established that AN depletes glutathione in the context of toxic liver injury, we hypothesize a similar systemic depletion may broadly impair the hepatic detoxification of reactive drug metabolites, lowering the hypersensitivity threshold. This metabolic instability creates a vulnerable phenotype for idiosyncratic drug reactions. (Zenger et al., 2004)
Conclusion
This case illustrates a severe presentation of lamotrigine-induced SJS triggered by a grossly inappropriate dose escalation and complicated by Atypical Anorexia Nervosa. It highlights that physiological alterations and polypharmacy act as silent risk factors for SCARs. Clinicians must exercise strict adherence to slow lamotrigine titration schedules, recognizing rapid escalation as a critical, preventable prescribing error.
Ethical Approval and Consent
Written informed consent was obtained from the patient for the publication of this case report and any accompanying clinical images.
References:
Benet LZ, Hoener BA. Changes in plasma protein binding have little clinical relevance. Clin Pharmacol Ther 2002; 71: 115-121.
Chung WH, Hung SI. Recent advances in the genetics and immunology of Stevens-Johnson syndrome and toxic epidermal necrolysis. J Dermatol Sci 2012; 66: 190-196.
Edinoff AN, Nguyen LH, Fitz-Gerald MJ, Crane E, Lewis K, Pierre SS, et al. Lamotrigine and Stevens-Johnson Syndrome prevention. Psychopharmacol Bull 2021; 51: 96-114.
Harr T, French LE. Toxic epidermal necrolysis and Stevens-Johnson syndrome. Orphanet J Rare Dis 2010; 5: 39.
McCormack M, Alfirevic A, Bourgeois S, et al. HLA-A*3101 and carbamazepine-induced hypersensitivity. N Engl J Med 2011; 364: 1134-1143.
Micheletti RG, Chiesa-Fuxench Z, Noe MH, et al. Stevens-Johnson Syndrome/Toxic Epidermal Necrolysis: A Multicenter Retrospective Study. J Invest Dermatol 2018; 138: 2315-2321.
Mockenhaupt M, Messenheimer J, Tennis P, Schlingmann J. Risk of Stevens-Johnson syndrome and toxic epidermal necrolysis in new users of antiepileptics. Neurology 2005; 64: 1134-1138.
Naranjo CA, Busto U, Sellers EM, Sandor P, Ruiz I, Roberts EA, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther 1981; 30: 239-245.
Roujeau JC, Kelly JP, Naldi L, Rzany B, Stern RS, Anderson T, et al. Medication use and the risk of Stevens-Johnson syndrome or toxic epidermal necrolysis. N Engl J Med 1995; 333: 1600-1607.
Schwartz RA, McDonough PH, Lee BW. Toxic epidermal necrolysis: Part I. Introduction, classification, clinical features, etiology, and immunopathogenesis. J Am Acad Dermatol 2013; 69: 173.e1-173.e13.
Spina E, de Leon J. Metabolic drug interactions with newer antipsychotics: a comparative review. Basic Clin Pharmacol Toxicol 2007; 100: 4-22.
Stern RS. Recurrence of Stevens-Johnson syndrome and toxic epidermal necrolysis. J Am Med Assoc 2014; 312: 1590-1591.
Winston AP. The clinical biochemistry of anorexia nervosa. Ann Clin Biochem 2012; 49: 132-143.
Zenger F, Russmann S, Junker E, Wuthrich C, Bui MH, Lauterburg BH. Decreased glutathione in patients with anorexia nervosa: Risk factor for toxic liver injury? Eur J Clin Nutr 2004; 58: 238-243.
|



