FRONTIERS IN MEDICAL CASE REPORTS - Volume 7; Issue 3, (May-Jun, 2026)
Pages: 01-17
Print Article
Download XML Download PDF
Clinical Improvement in Postural Orthostatic Tachycardia Syndrome (POTS) Following Prolotherapy and Cervical Curve Correction: 4 Case Series of Patients with Hypermobile Ehlers-Danlos Syndrome (hEDS) and Suspected Cervicovagopathy
Author: Hauser R, Griffiths M, Rawlings B, Deadwyler H, Matias D, Smith J, Foster D
Category: Medical Case Reports
Abstract:
Background: Postural orthostatic tachycardia syndrome (POTS) is common in hypermobile Ehlers-Danlos syndrome (hEDS). This retrospective case series presents 4 cases of improvement in POTS symptoms in patients with hEDS following combined intervention of prolotherapy and at-home cervical curve correction protocols. Objective: We present the clinical histories and objective test findings of 4 patients with hEDS and POTS who underwent prolotherapy and cervical curve correction protocols to treat ligamentous cervical instability, with observed objective and subjective clinical changes, including improvement of POTS symptoms. Results: All 4 patients had significant cervical structural abnormalities: ligamentous cervical instability at the atlantoaxial joint and forward head posture identified with digital motion x-ray (DMX), and decreased vagus nerve and internal jugular vein (IJV) cross-sectional areas (CSA) identified with ultrasound. Following prolotherapy treatments and an individualized curve correction protocol, all 4 patients demonstrated objective changes and self-reported improvement of their POTS symptoms. Conclusion: These cases describe patients with hEDS and POTS who demonstrate radiologically identified ligamentous cervical instability and structural forward head posture, and ultrasound-identified decreased vagus nerve and IJV CSAs. Prolotherapy and cervical curve correction were associated with improvements in objective test measures, as well as meaningful self-reported improvement in POTS symptoms. These findings raise the possibility that reduced vagus nerve CSA may contribute to POTS symptoms in the context of structural cervical spine abnormalities.
Keywords: Postural Orthostatic Tachycardia Syndrome, Hypermobile Ehlers-Danlos Syndrome, Ligamentous Cervical Instability, Case Report, Vagus Nerve Degeneration, Dysautonomia, Cervical Lordosis, Prolotherapy
Full Text:
Introduction
Postural orthostatic tachycardia syndrome (POTS), a complex, heterogeneous, and often debilitating disorder, is a prominent comorbidity in patients with hEDS (Wallman et al., 2014). POTS is characterized by an increase in heart rate by more than 30 beats per minute within 10 minutes of standing while maintaining (or a slight increase in) blood pressure (Schondorf and Low, 1993). Associations between POTS and hEDS have been documented in many studies and continue to be explored, yet potential underlying mechanisms remain elusive and the high overlap of the 2 conditions remains an enigma (Gazit et al., 2003; Kanjwal et al., 2010).
Around 30-40% of patients with hEDS are thought to have POTS, 150 times more than the general population (Roma et al., 2018; Hertel et al., 2024). Clinically, POTS and hEDS are both more frequently seen in women, with previous studies showing 70-90% and up to 91% of patients being female in POTS and hEDS, respectively (Shaw et al., 2019; Glayzer et al., 2024; Wilson et al., 2026). Common treatments for POTS include increasing fluid and salt intake, compression garments, graded exercise, and various medications aimed at reducing heart rate, increasing blood volume, supporting blood pressure, and reducing sympathetic overactivity (Benarroch, 2012; Fu and Levine, 2018; Miller and Raj, 2018; Raj et al., 2022). There is no single treatment that works for everyone who has POTS, nor a proven permanent cure, though it is noted that some patients spontaneously recover (Fedorowski, 2019; Vasavada et al., 2023).
While the pathophysiology of POTS is complex, and many mechanisms have been described, it is primarily defined as an autonomic dysfunction—specifically, sympathetic overdrive or decreased parasympathetic activity, which can cause higher heart rate, reduced heart rate variability, and impaired blood pressure regulation (reduced cardiovagal activity), indicating underactivity of the vagus nerves (Muenter Swift et al., 2005; Benarroch, 2012). The primary mechanisms of heightened sympathetic activity believed to contribute to the orthostatic intolerance in POTS include volume dysregulation, impaired vasoconstriction, and hyperadrenergic states (Stewart, 2012; Zhao and Tran, 2026). Cervical spine instability and structural abnormalities have been reported in patients with hEDS and may have implications for adjacent neurovascular structures, yet their relationship to autonomic dysfunction in POTS remains poorly characterized.
This paper presents 4 cases in which patients with hEDS experienced marked improvement of symptoms attributed to POTS following cervical spine treatment with prolotherapy and cervical curve-corrective postural therapies. In these 4 cases, treatment was directed at ligamentous cervical instability and structural forward head posture (documented by digital motion x-ray [DMX]) with the aim of potentially reducing stretch or compression of the internal jugular veins (IJVs) and vagus nerves (documented by ultrasound imaging of the carotid sheath).
Methods
Background and Diagnostics: All 4 cases presented in this paper involve patients with hEDS who sought evaluation at an outpatient neck center to assess their cervical spine as a potential contributing factor to their clinical presentation, including POTS as a primary complaint. POTS was recorded as a pre-existing diagnosis based on patient report at intake. Details regarding the diagnosing clinician, criteria used, and results of formal autonomic testing were not available for independent review in this study. Standard diagnostic criteria consist of orthostatic intolerance, increased heart rate of at least 30 beats upon standing, and characteristic symptoms of dizziness and/or lightheadedness upon going from supine to upright position (Fedorowski, 2019). Every patient noted previous POTS treatments with limited improvement; details of previous treatment, however, were not included in this study.
All 4 patients underwent clinical testing involving upright digital motion x-ray (videofluoroscopy) and/or cone beam CT of the cervical spine in various positions, including open-mouth views with lateral flexion to evaluate ligamentous upper cervical stability of the C1-C2 (atlantoaxial) joint and forward head posture, documented by measuring the C6-atlas interval (C6AI), the distance between the posterior aspect of the C6 vertebral body and the anterior arch of C1 (atlas) (Fig. 1). Ultrasounds of the carotid sheath were performed to document the CSAs of the internal jugular veins and vagus nerves in various positions (Fig. 2).
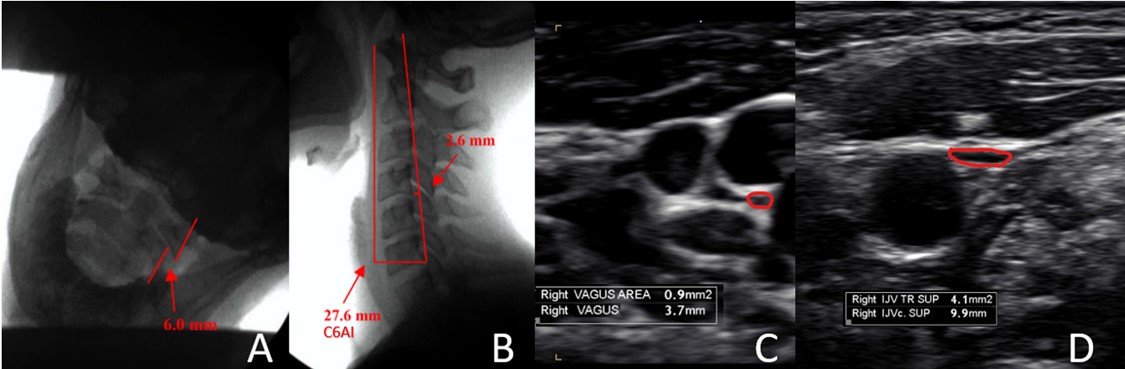
Figure 1: A. DMX showing severe ligamentous cervical instability (LCI) at C1-C2, open-mouth lateral flexion. B. DMX sagittal view with C6AI measurement. C. Ultrasound vagus nerve measurement. D. Ultrasound IJV measurement.
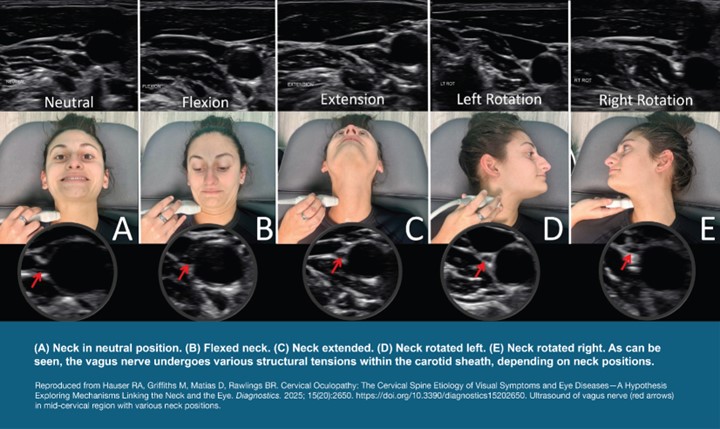
Figure 2: Ultrasound measurements of the vagus nerve in various positions.
Digital motion x-ray (DMX) and ultrasound measurements were performed at a single outpatient center by certified radiology technologists or medical ultrasonographers following standard clinical procedures. Patient positioning was replicated as closely as possible to promote consistency between baseline and follow-up. Due to the retrospective nature of the study, measurements were done for clinical, not research, purposes, minimizing potential bias. Assessors were not blinded to treatment stage or clinical status, as imaging and analysis were performed as part of routine clinical care. Ultrasound measurements obtained in the supine position with a neutral head and neck alignment were included in the analysis to allow for standardized comparisons between baseline and follow-up. The supine position was selected as a standardized reference given the reduced cervical strain and relative expected maximization of IJV cross-sectional area (Czyżewska et al., 2015).
DMX provides a high degree of accuracy for identifying vertebral instability, as it allows for real-time motion imaging in the upright position, which can aid in the diagnosis of pathology that static imaging in the supine position could miss (Freeman et al., 2020). The open-mouth lateral bending position used to evaluate LCI C1-C2 demonstrates good-to-excellent inter-examiner reliability, with intraclass correlation coefficient ranging from 0.56-0.97. Ultrasound evaluation of the vagus nerves’ cross-sectional areas at the level of the thyroid gland (mid-cervical region, C5) has good inter-rater agreement, with intraclass correlation coefficients from 0.72-0.97 for inter-rater reliability (Belau and Pelz, 2017; Dong et al., 2024). Ultrasound measurement of the internal jugular veins’ cross-sectional areas demonstrates good-to-high inter-rater reliability, with intraclass correlation coefficients over 0.82-0.93 (Donahue et al., 2009; Parenti et al., 2019; Chayapinun et al., 2024).
Assessing the cross-sectional area of the IJVs at the mid-neck (C4–C5) and upper neck (C1–C2) using B-mode ultrasound across seated and supine positions, and with varying head and neck orientations, can help identify the presence and extent of compression or abnormalities (Hauser et al., 2026). IJV CSA was measured using B-mode ultrasound with manual tracing of the central luminal boundary at a predefined anatomical level, the lateral mass of C1. Normal IJV CSA in the supine position is >90-100 mm2 (Tartière et al., 2009; Yoon et al., 2013). The vagus nerve was identified within the carotid sheath and measured at anatomical level C4-C5. The most common location of the vagus nerve in the mid-neck is lying posterior to the IJV and lateral to the carotid artery, with normal vagus nerve CSA being 2-3 mm2 (Abdelnaby et al., 2022; Ahn et al., 2022).
Common diagnosis in all 4 cases: Each patient was found to have significant ligamentous cervical instability of the C1-C2 atlantoaxial joint, forward head posture documented by C6-atlas interval, and decreased IJV and vagus nerve CSAs.
Treatment: The primary intervention consisted of multiple courses of prolotherapy treatments over a time span of at least 6 months to address the ligamentous upper cervical instability (Table 1). Injection sites and technique were individualized to each patient’s clinical presentation and followed standard clinical practice. All patients were additionally recommended individualized cervical curve correction protocols based on the lateral neck view of the digital motion x-ray (videofluoroscopy) and clinical test results. Cervical curve correction protocols contained customized recommendations for exercises to help re-establish the cervical lordotic curve, as well as lifestyle recommendations such as ergonomic work postures and resting positions that promote improved internal jugular vein drainage. These protocols were intended as an adjunct to in-clinic treatment, but adherence, frequency, and execution were not formally monitored nor recorded. The goal of the combined treatment was to gain stability in the cervical spine, improve cervical spine sagittal alignment, and relieve pressure or tension on the internal jugular veins and vagus nerves, which were hypothesized to be contributing to clinical presentation. Other concurrent therapies, if performed, were not systematically recorded.
Results
All 4 patients reported significant improvement in their POTS symptoms. Across all 4 patients, the average change in total LCI C1-C2 was -3.3 mm, with mean 6.5 treatments per patient (Table 1). for demographics. On follow-up testing, each patient showed improvement in ligamentous cervical instability at C1-C2, cervical curve sagittal alignment (forward head posture measured as C6-atlas interval), IJV CSA, and vagus nerve CSA (average change of -3.3 mm, -16.5 mm, +29.6 mm2, and +1.8 mm2, respectively). (Table 2) Adherence to the prescribed home-based protocol could not be assessed.
Table 1: Number of prolotherapy treatments between initial clinical testing and follow-up testing, and subjective improvement of postural orthostatic tachycardia syndrome.
Table 2: Initial and post-treatment objective cervical spine and neurovascular measurements in 4 clinical cases of patients with hEDS and POTS.
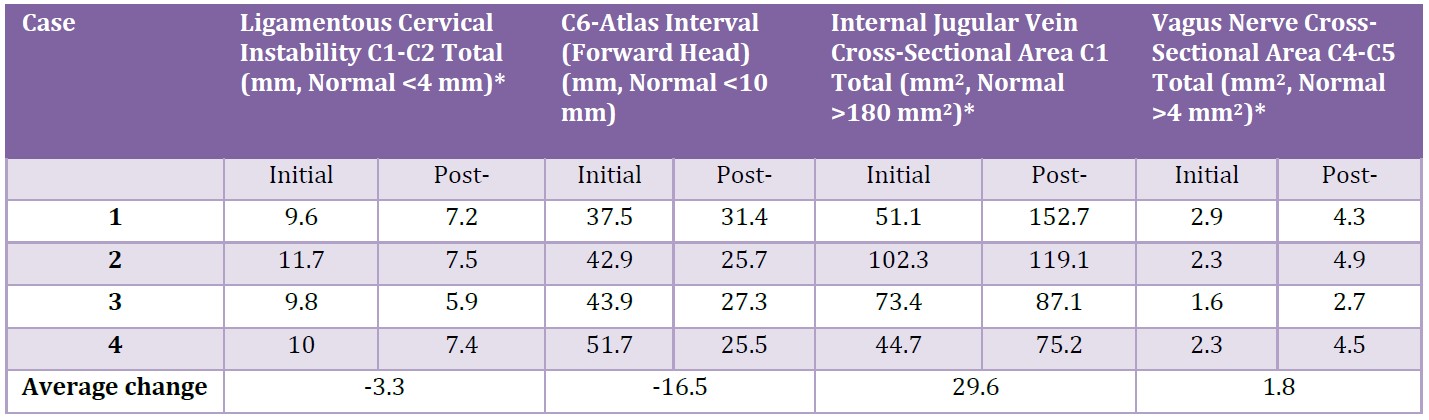
* Data are collected unilaterally but reported here as bilateral totals (except for C6-atlas interval) due to clinical bilateral symmetry, and to streamline results.
Description of Cases
Patient # 1
Clinical notes: A 35-year-old female presented to Caring Medical in 2023 with debilitating neck pain, dizziness, headaches/migraines, jaw pain, visual disturbances, brain fog, and declined quality of life. She had previous diagnoses of hEDS and POTS. Changing positions triggered symptoms. When she bent over, she experienced dizziness, nausea, and body temperature changes. The patient reported having tightness and grinding throughout her neck that radiates to the jaw. She had a long history of frequent dislocation and subluxation in her neck, bilateral shoulders/scapula, elbows, ribs, hips, and left knee. The patient believed that most of her symptoms were coming from cervical instability, as her neck clicked and popped a lot with motions. Previous treatment included physical therapy.
Treatments: The patient received 9 prolotherapy treatments over the course of 15 months and was recommended to have an individualized cervical curve correction program to do at home in between treatments.
Outcome: The patient’s LCI C1-C2 significantly improved, vagus nerve CSAs increased by 48%, and IJV CSA at C1 tripled, going from 51.1 mm2 total to 152.7 mm2 total. In addition to significant objective improvement (Table 1), the patient reports her visual disturbances and brain fog have resolved and her POTS symptoms have improved by 90%.
Patient # 2:
Clinical notes: A 39-year-old female sought care at Caring Medical in 2024, reporting a constellation of escalating symptoms. Her chief complaints included drop attacks, significant brain fog, chronic dizziness, persistent nausea, debilitating fatigue, migraines, and severe insomnia. She also experienced marked autonomic dysfunction, manifesting as fluctuating heart rates (tachycardia and bradycardia) and an inability to regulate her blood pressure. The patient's history is notable for several concussions, often a consequence of blackouts. She also reported a motor vehicle accident. In 2019, she received diagnoses of hEDS and POTS. Her symptoms progressively worsened over time, and she had begun to experience new, additional issues. Through her own online research, the patient posited that cervical instability, a sequela of her hEDS compounded by various accidents over the years, was the underlying cause of many of her symptoms.
Treatments: The patient received 7 prolotherapy treatments over the course of 6 months and was recommended to have an individualized cervical curve correction program to do at home in between treatments.
Outcome: The patient’s follow-up testing documented significant improvement in LCI C1-C2, going from 11.7 mm to 7.5 mm total, significant C6AI (forward head) improvement from 42.9 mm to 25.7 mm, and vagus nerve CSAs more than doubled, from 2.3 mm2 to 4.9 mm2 total. In addition to significant objective improvement (Table 1), the patient reported her symptoms are 70% improved, including those attributed to POTS. The drop attacks reduced from a couple of times a week to once a month. She indicated that her overall energy has improved, her migraines have resolved, and she is now able to keep up with her life.
Patient # 3:
Clinical notes: A 21-year-old female presented to Caring Medical in 2024 with multifactorial symptoms that included orthostatic intolerance (dizziness, lightheadedness, racing heart, and palpitations upon standing), cognitive dysfunction (brain fog, impaired concentration, and difficulty with memory), cervicalgia, ocular pressure, and photophobia. Her history included a diagnosis of POTS and chronic fatigue syndrome (CFS) from a dysautonomia specialist, and she also met the criteria for hEDS. She reported daily drop attacks, exertional intolerance—specifically an inability to exercise due to presyncope or syncopal episodes—and cognitive deficits, including impaired focus, concentration, and memory, all associated with severe fatigue. Extensive diagnostic testing revealed that the patient had ligamentous upper cervical instability, specifically severe C1-C2 instability (worse on the left) and rotational instability. Additionally, she presented with bilateral elongated styloid processes, severe temporomandibular joint instability, bilateral internal jugular vein compression, bilateral degeneration of the vagus nerves, and swollen optic nerves.
Treatments: The patient underwent 4 prolotherapy treatments over the course of 10 months and was recommended to have an individualized cervical curve correction program to do at home in between treatments.
Outcome: This patient demonstrated a notable reduction in LCI C1-C2 from 9.8 mm to 5.9 mm total, and the C6-atlas interval improved from 43.9 mm to 29.2 mm. Her vagus nerves increased by 68%. In addition to significant objective improvement (Table 1), the patient reported a 50% overall improvement in her symptoms. She notes feeling better waking up, and her fatigue is no longer debilitating throughout the day. She further states that the treatments have enhanced her ability to manage stress. She was also preparing to graduate from college, indicating an improved capacity for daily activities and future planning.
Patient # 4:
Clinical notes: A 20-year-old female was referred to Caring Medical in 2023 for a second opinion regarding hEDS and suspected cervical instability. The patient, a former dancer, had been homebound for most of 2022-23, entirely stopping school due to her symptoms, though she hoped to return before graduating. She reported no trouble walking but used a scooter for long distances. The patient's onset of complex symptoms began over 10 years earlier with chronic neck pain. She received an hEDS diagnosis at age 16 and bilateral trigeminal neuralgia at age 17. Despite numerous prior imaging studies (e.g., MRIs, x-rays, and CT scans) showing only a straight neck, an upright MRI shortly before coming to this clinic demonstrated instability in the lower cervical spine and ligament laxity, leading her to suspect craniocervical instability. Her 3 worst symptoms were unrelenting trigeminal neuralgia, which was unresponsive to any therapies she had tried in the previous year; debilitating, constant neck pain described as a burning, pressure, and stabbing sensation, accompanied by a deep clicking/popping of her vertebrae; and extreme, profuse sweating all over her body, even in 70°F still air. She believed the profuse sweating was related to a POTS diagnosis which had been noted for several years. The neck pain was constant, sometimes worse on the right, and was felt in both the C1-C2 and lower neck regions, radiating down to her shoulders with positive crepitus and reported episodes of the neck going "out of place." The trigeminal neuralgia pain was described as burning and electric, almost constant across her entire face, eyes, head, and jaw. Associated symptoms included headaches, head pressure, light sensitivity, and a feeling of pressure/pain in the eyes. She also reported ringing (tinnitus), pain, and a full feeling in her ears, which are triggered by loud noises. The patient experienced dizziness, balance issues, brain fog, difficulty keeping her head up, and occasional numbness and tingling in her arms and legs. She had a history of a temporomandibular joint disorder diagnosis and reported jaw pain. Of significant concern, the patient experienced a drop attack a few days prior to her first appointment at this clinic, falling to the floor shortly after getting out of bed, which she did not feel coming on, unlike what she typically anticipated with POTS symptoms.
The patient also had diagnoses of complex PTSD and panic disorder stemming from her chronic medical history. She has reported feeling stable and notes mental improvements since seeing a chronic pain psychiatrist over the past year, a period during which she also saw a chiropractor. She had visited many specialists without experiencing any significant improvement, including a geneticist, neurologist, cardiologist, GI specialist, and sleep expert, and had undergone various treatments, including medications, ketamine infusions, and an epidural for low back pain (which were unhelpful). She is currently followed by a chronic pain psychiatrist, pediatrician, gastroenterologist, neurologist, and a cardiologist.
Treatment: The patient underwent 4 prolotherapy treatments over the course of 11 months and was recommended to have individualized cervical curve correction program to do at home in between treatments.
Outcome: This patient demonstrated significant improvement in LCI C1-C2, C6AI, IJV CSA, and vagus nerve CSA. Her forward head posture (C6AI measurement) decreased by half, from 51.7 mm to 25.5 mm, and her vagus nerve CSAs nearly doubled, going from 2.3 mm2 to 4.5 mm2 total. In addition to significant objective improvement (Table 1), the patient reported feeling good enough to enter college—a major improvement from being homebound. Overall, she reports an 80% improvement in her symptoms, including brain function, mental health issues, trigeminal neuralgia, walking ability, temperature regulation, and symptoms associated with POTS.
Discussion
Each of these 4 cases demonstrates improvement in ligamentous upper cervical instability of C1-C2 atlantoaxial joints, structural forward head posture in the sagittal plane, and cross-sectional areas of the vagus nerves and internal jugular veins. In addition to the objective improvement following treatment targeting the cervical spine, all 4 cases also reported significant subjective improvement in their overall condition, including symptoms attributed to POTS. These observations suggest a potential association between improvements in ligamentous cervical stability and cervical lordosis following prolotherapy, and improvements in POTS symptoms in patients with hEDS. It was clinically hypothesized that the patients’ POTS symptoms could be due to cervicovagopathy, a recently described term to denote vagus nerve degeneration from cervical structural changes, including cervical dysstructure (breakdown of the neck curve) and ligamentous cervical instability (Hauser et al., 2024) (Fig. 3). It is mechanistically plausible that the observed improvement in POTS symptoms noted in these 4 cases may be associated with changes in cervical structure and reduced vagus nerve irritation (stretch, compression, or degeneration), with concurrent increases in vagus nerve CSA—it has been documented that vagus nerve CSA correlates with parasympathetic dysfunction, including heart rate and tilt test parameters (Huckemann et al., 2023).
Vagal withdrawal is a notable etiological feature in many POTS cases, and is linked to cardiovagal baroreflex dysfunction, which can cause blood pressure volatility and resultant elevated heart rate (Lecheler et al., 2021; Stewart et al., 2021; Chakraborty et al., 2023). At the crux of some cases of POTS may therefore be cardiovagal baroreflex dysfunction, elevating heart rate and causing low heart rate variability. POTS is one of the more debilitating forms of dysautonomia, associated with a variety of symptoms, including dizziness, lightheadedness, fainting, temperature dysregulation, joint pain and instability, fatigue, and more (Trout et al., 2025). More than 50% of POTS patients experience orthostatic headaches (Khurana and Eisenberg, 2011). POTS symptoms of cerebral hypoperfusion typically improve with lying down, but symptoms of sympathetic hyperactivity may persist, including heart palpitations, dizziness or fainting when upright, blurred vision, weakness in the legs, chest pain, nausea, shortness of breath, fatigue, and difficulty concentrating (Nwazue and Raj, 2013; Deb et al., 2015).
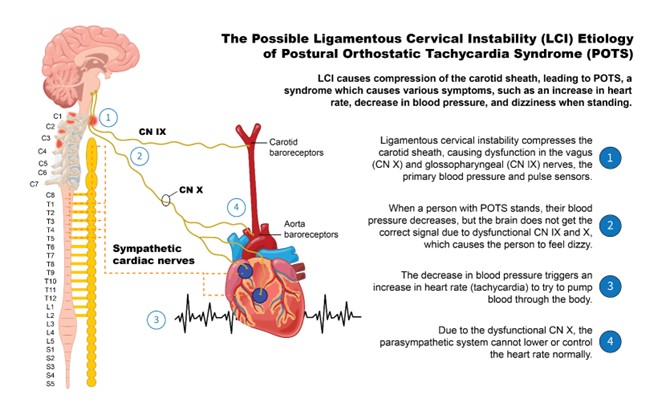
Figure 3: The Possible Ligamentous Cervical Instability (LCI) Etiology of Postural Orthostatic Tachycardia Syndrome (POTS).
With ligamentous cervical instability, the cervical curve can break down, and the atlas can shift forward in 3-D space (structural forward head posture) (Hauser et al., 2024). The forward shift of the atlas can cause compression of the carotid sheath, including the vagus nerves and internal jugular veins (Fig. 4). Cervicovagopathy—degeneration, irritation, or functional impairment of the vagus nerve caused by structural neck issues, was clinically predicted to be the underlying cause of automimic dysfunction, causing POTS in these 4 patients, with the vagus nerve CSA being a notable objective improvement in all 4 cases (Hauser et al., 2025). The vagus nerves are the primary inhibitors of the sympathetic nervous system, traversing the neck just anterior to the cervical vertebrae within the carotid sheath. The vagus nerve ganglia, nodose (inferior) and jugular (superior), lie just anterior to the posterior arch of the atlas (Hauser et al., 2026). Cervicovagopathy can be caused by documented ligamentous cervical instability, both of which showed significant improvement following prolotherapy and curve-correcting treatment (Hauser et al., 2025).
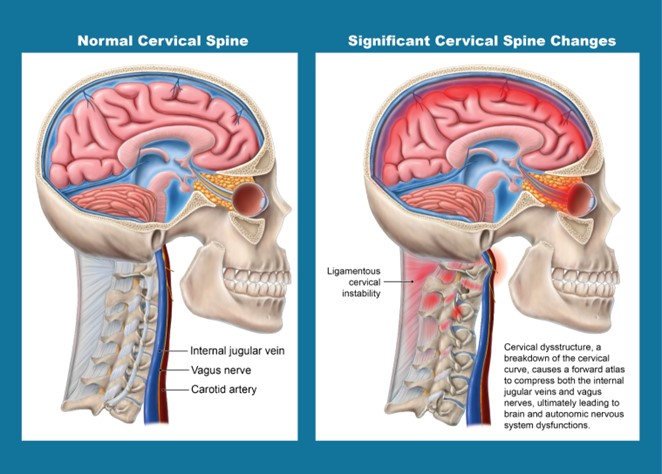
Figure 4: Potential Ligamentous Cervical Instability Etiology of Autonomic Nervous System Dysfunction.
Prolotherapy, a regenerative injection therapy directed at the posterior ligament complex of the neck, especially the C1-C2 capsular ligaments, targets the prominent ligamentous cervical instability. Prolotherapy has been shown to help strengthen ligaments and has been used successfully for cervical conditions, including chronic pain and cervical instability (Centeno et al., 2005; Hauser et al., 2015). To treat the structural forward head posture identified by C6AI measurements, patients were educated on ergonomics and proper posture and recommended various exercises and therapies to do at home, including lying on a cervical orthotic device, the Denneroll®. Following combined treatment to the cervical spine, objective changes were observed in the CSAs of the vagus nerves and internal jugular veins.
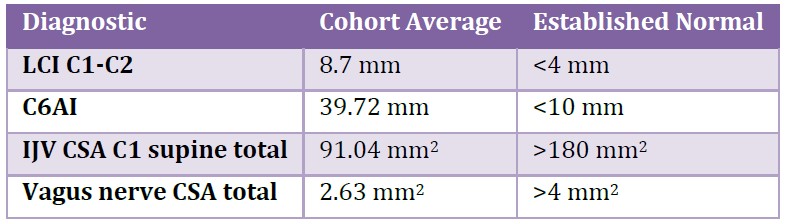
Patients with hEDS have generalized ligament laxity, predisposing some of them to develop ligamentous cervical instability and loss of normal cervical lordosis (Gensemer et al., 2021). This case series illustrates a recurring clinical pattern observed at this outpatient neck center. It is a common finding in this clinic to see cervical structural pathologies (LCI and forward head) leading to compression of the carotid sheath, seen as decreased vagus nerve and IJV CSAs. Similar findings of ligamentous cervical instability at C1-C2, structural forward head posture, and decreased IJV and vagus nerve CSAs were observed in a larger clinical cohort of similar profile, 20 patients with hEDS and POTS from the same clinic as these 4 cases, who were evaluated using the same diagnostic approach, lending contextual support to the patterns described in the present cases (Table 3).
Table 3: Mean diagnostic results of 20 patients with hEDS and POTS at an outpatient neck clinic.
To our knowledge, this is the first report of objective improvement in vagus nerve CSA alongside patient-reported improvement in POTS symptoms in patients with hEDS following treatment targeting ligamentous cervical instability and cervical spine dysstructure. Supporting research already links the vagus nerves to POTS, with reduced vagal control emphasized in the neuropathic subtype of POTS, and vagus nerve stimulation thought to decrease POTS symptoms by improving vagal tone (Jacob et al., 2019). Additionally, low vagus nerve CSA has correlated with poor parasympathetic function, and parasympathetic dysfunction is commonly found in POTS patients (Celletti et al., 2017; Huckemann et al., 2022). This case series raises the possibility that cervical spine factors may represent a potential contributing mechanism in a subset of patients with POTS, possibly involving effects on the vagus nerves.
The observations in the 4 cases presented in this paper suggest a possible association between treatment targeting ligamentous cervical instability and cervical lordosis and improvement in POTS symptoms in patients with hEDS. The intervention included both prolotherapy and an unmonitored home-based cervical rehabilitation program, limiting the ability to isolate the independent effect of prolotherapy or curve corrective protocols. Adherence to the home program, as well as potential co-interventions, were not tracked. As such, this case series is intended to be hypothesis-generating rather than establishing causality. The retrospective nature of the design introduces inherent selection bias; We selected 4 cases with favorable treatment responses to enable consideration of potential mechanisms and therapeutic approaches. These observations support the hypothesis that cervicovagopathy could be a potential contributing factor in a subset of patients with POTS and hEDS. Should these findings be confirmed in larger prospective studies, a subset of patients with POTS and hEDS may benefit from the development of novel treatment options.
Conclusion
These cases describe patients with hEDS and POTS who demonstrate radiologically identified ligamentous cervical instability and structural forward head posture, and ultrasound-identified decreased vagus nerve and IJV CSAs. Prolotherapy and cervical curve correction were associated with improvements in objective test measures, as well as meaningful self-reported improvements in POTS symptoms. These findings raise the possibility that reduced vagus nerve CSA may contribute to POTS symptoms in the context of structural cervical spine abnormalities. While the interventions were associated with improved cervical spine structure and stability, the findings suggest that improved vagus nerve CSA may influence vagal function and could potentially be associated with symptom improvement in patients with POTS in the hEDS population. The cervical spine and neurovascular patterns observed in these 4 cases may provide a framework for future, controlled, hypothesis-driven studies.
Ethical Approval and Consent: Written informed consent was obtained from the patients for the publication of this case series.
Abbreviations: IJV: Internal jugular vein; CSA: Cross-sectional area; LCI: Ligamentous cervical instability; C6AI: C6-atlas interval; DMX: Digital motion x-ray; hEDS: Hypermobile Ehlers-Danlos syndrome; MRI: Magnetic resonance imaging; POTS: Postural orthostatic tachycardia syndrome.
Acknowledgements: We would like to thank Ashley Watterson for her contribution of data analysis, and Kerstyn Gay for her contribution to the graphic designs.
References:
Abdelnaby R, Elsayed M, Mohamed KA, Dardeer KT, Sonbol YT, ELgenidy A, Barakat MH, NasrEldin YK, Maier A. Sonographic Reference Values of Vagus Nerve: A Systematic Review and Meta-analysis. J Clin Neurophysiol 2022; 39: 59-71.
Ahn D, Lee GJ, Sohn JH, Kim JK. Prevalence and characteristics of vagus nerve variations on neck ultrasonography. Ultrasonography 2022; 41: 124-130.
Belau E, Pelz J, Weise D. P 25 Reference values for the cross-sectional area of the vagus nerve in healthy subjects–a high-resolution ultrasound study. Clinical Neurophysiology 2017; 128: e339.
Benarroch EE. Postural tachycardia syndrome: a heterogeneous and multifactorial disorder. Mayo Clin Proc 2012; 87: 1214-1225.
Celletti C, Camerota F, Castori M, Censi F, Gioffrè L, Calcagnini G, Strano S. Orthostatic Intolerance and Postural Orthostatic Tachycardia Syndrome in Joint Hypermobility Syndrome/Ehlers-Danlos Syndrome, Hypermobility Type: Neurovegetative Dysregulation or Autonomic Failure? Biomed Res Int 2017; 2017: 9161865.
Centeno CJ, Elliott J, Elkins WL, Freeman M. Fluoroscopically guided cervical prolotherapy for instability with blinded pre and post radiographic reading. Pain Physician 2005; 8: 67-72.
Chakraborty P, Farhat K, Morris L, Whyte S, Yu X, Stavrakis S. Non-invasive Vagus Nerve Simulation in Postural Orthostatic Tachycardia Syndrome. Arrhythm Electrophysiol Rev 2023; 12: e31.
Chayapinun V, Koratala A, Assavapokee T. Seeing beneath the surface: Harnessing point-of-care ultrasound for internal jugular vein evaluation. World J Cardiol 2024; 16: 73-79.
Czyżewska D, Krysiuk K, Dobrzycki K, Ustymowicz A. Ultrasound assessment of the jugular and vertebral veins in healthy individuals: selected physiological aspects and morphological parameters. J Ultrason 2015; 15: 267-273.
Deb A, Morgenshtern K, Culbertson CJ, Wang LB, Hohler AD. A survey-based analysis of symptoms in patients with postural orthostatic tachycardia syndrome. Proc (Bayl Univ Med Cent) 2015; 28: 157-159.
Donahue SP, Wood JP, Patel BM, Quinn JV. Correlation of sonographic measurements of the internal jugular vein with central venous pressure. Am J Emerg Med 2009; 27: 851-855.
Dong S, Shen B, Jiang X, Zhu J, Zhang H, Zhao Y, Chen Y, Li D, Feng Y, Chen Y, Pan Y, Han F, Liu B, Zhang L. Comparison of vagus nerve cross-sectional area between brain-first and body-first Parkinson's disease. NPJ Parkinsons Dis 2024; 10: 231.
Fedorowski A. Postural orthostatic tachycardia syndrome: clinical presentation, aetiology and management. J Intern Med 2019; 285: 352-366.
Freeman MD, Katz EA, Rosa SL, Gatterman BG, Strömmer EMF, Leith WM. Diagnostic Accuracy of Videofluoroscopy for Symptomatic Cervical Spine Injury Following Whiplash Trauma. Int J Environ Res Public Health 2020; 17: 1693.
Fu Q, Levine BD. Exercise and non-pharmacological treatment of POTS. Auton Neurosci 2018; 215: 20-27.
Gazit Y, Nahir AM, Grahame R, Jacob G. Dysautonomia in the joint hypermobility syndrome. Am J Med 2003; 115: 33-40.
Gensemer C, Burks R, Kautz S, Judge DP, Lavallee M, Norris RA. Hypermobile Ehlers-Danlos syndromes: Complex phenotypes, challenging diagnoses, and poorly understood causes. Dev Dyn 2021; 250: 318-344.
Glayzer JE, Bray BC, Kobak WH, Steffen AD, Schlaeger JM. Lack of Diversity in Research on Females with Ehlers-Danlos Syndromes: Recruitment Protocol for a Quantitative Online Survey. JMIR Res Protoc 2024; 13: e53646.
Hauser RA, Griffiths M, Watterson A, Matias D, Rawlings BR. Characterizing Initial Cervical Spine and Neurovascular Findings in 84 Consecutive Patients with Hypermobile Ehlers-Danlos Syndrome: A Retrospective Study. J Clin Med 2026; 15: 2212.
Hauser RA, Matias D, Rawlings B. The ligamentous cervical instability etiology of human disease from the forward head-facedown lifestyle: emphasis on obstruction of fluid flow into and out of the brain. Front Neurol 2024; 15: 1430390.
Hauser RA, Matias D, Rawlings BR. Cervicovagopathy: ligamentous cervical instability and dysstructure as a potential etiology for vagus nerve dysfunction in the cause of human symptoms and diseases. Frontiers in Neurology 2025; 16: 1572863.
Hauser RA, Steilen D, Gordin K. The biology of Prolotherapy and its application in clinical cervical spine instability and chronic neck pain: a retrospective study. European Journal of Preventive Medicine 2015; 3: 85-102.
Hertel AK, Black WR, Lytch A, Cramer E, Malloy Walton L, Jones JT. Cardiovascular, autonomic symptoms and quality of life in children with hypermobile Ehlers-Danlos syndrome. SAGE Open Med 2024; 12: 20503121241287073.
Huckemann S, Mueller K, Averdunk P, Kühn E, Hilker L, Kools S, Scholz L, Bulut Y, Brünger J, Fiegert S, Grüter T, Fisse AL, Motte J, Yoon MS, Gold R, Schneider-Gold C, Tönges L, Pitarokoili K. Vagal cross-sectional area correlates with parasympathetic dysfunction in Parkinson's disease. Brain Commun 2023; 5: fcad006.
Jacob G, Diedrich L, Sato K, Brychta RJ, Raj SR, Robertson D, Biaggioni I, Diedrich A. Vagal and Sympathetic Function in Neuropathic Postural Tachycardia Syndrome. Hypertension 2019; 73: 1087-1096.
Kanjwal K, Saeed B, Karabin B, Kanjwal Y, Grubb BP. Comparative clinical profile of postural orthostatic tachycardia patients with and without joint hypermobility syndrome. Indian Pacing Electrophysiol J 2010; 10: 173-178.
Khurana RK, Eisenberg L. Orthostatic and non-orthostatic headache in postural tachycardia syndrome. Cephalalgia 2011; 31: 409-415.
Lecheler L, Hoffmann F, Tank J, Jordan J. Run vagus run: Cardiovagal baroreflex function and the postural tachycardia syndrome. Hypertension 2021; 77: 1245-1247.
Miller AJ, Raj SR. Pharmacotherapy for postural tachycardia syndrome. Auton Neurosci 2018; 215: 28-36.
Muenter Swift N, Charkoudian N, Dotson RM, Suarez GA, Low PA. Baroreflex control of muscle sympathetic nerve activity in postural orthostatic tachycardia syndrome. Am J Physiol Heart Circ Physiol 2005; 289: H1226-H1233.
Nwazue VC, Raj SR. Confounders of vasovagal syncope: postural tachycardia syndrome. Cardiol Clin 2013; 31: 101-109.
Parenti N, Scalese M, Palazzi C, Agrusta F, Cahill J, Agnelli G. Role of Internal Jugular Vein Ultrasound Measurements in the Assessment of Central Venous Pressure in Spontaneously Breathing Patients: A Systematic Review. J Acute Med 2019; 9: 39-48.
Raj SR, Fedorowski A, Sheldon RS. Diagnosis and management of postural orthostatic tachycardia syndrome. CMAJ 2022; 194: E378-E385.
Roma M, Marden CL, De Wandele I, Francomano CA, Rowe PC. Postural tachycardia syndrome and other forms of orthostatic intolerance in Ehlers-Danlos syndrome. Auton Neurosci 2018; 215: 89-96.
Schondorf R, Low PA. Idiopathic postural orthostatic tachycardia syndrome: an attenuated form of acute pandysautonomia? Neurology 1993; 43: 132-137.
Shaw BH, Stiles LE, Bourne K, Green EA, Shibao CA, Okamoto LE, Garland EM, Gamboa A, Diedrich A, Raj V, Sheldon RS, Biaggioni I, Robertson D, Raj SR. The face of postural tachycardia syndrome - insights from a large cross-sectional online community-based survey. J Intern Med 2019; 286: 438-448.
Stewart JM, Warsy IA, Visintainer P, Terilli C, Medow MS. Supine Parasympathetic Withdrawal and Upright Sympathetic Activation Underly Abnormalities of the Baroreflex in Postural Tachycardia Syndrome: Effects of Pyridostigmine and Digoxin. Hypertension 2021; 77: 1234-1244.
Stewart JM. Mechanisms of sympathetic regulation in orthostatic intolerance. J Appl Physiol (1985). 2012; 113: 1659-1668.
Tartière D, Seguin P, Juhel C, Laviolle B, Mallédant Y. Estimation of the diameter and cross-sectional area of the internal jugular veins in adult patients. Crit Care 2009; 13: R197.
Trout J, Vernino S, Codden R, Barson J, Laforge G, Billings A, Cortez M. Postural Tachycardia Syndrome (POTS) Symptom Burden: Frequency, Severity, and Impact on Activities of Daily Living (P1-7.009). InNeurology 2025 Apr 8 (Vol. 104, No. 7_Supplement_1, p. 2722). Hagerstown, MD: Lippincott Williams & Wilkins.
Vasavada AM, Verma D, Sheggari V, Ghetiya S, Chirumamilla PC, Kotak RA, Mahapatra SS, Patel T, Jain M. Choices and Challenges With Drug Therapy in Postural Orthostatic Tachycardia Syndrome: A Systematic Review. Cureus 2023; 15: e38887.
Wallman D, Weinberg J, Hohler AD. Ehlers-Danlos Syndrome and Postural Tachycardia Syndrome: a relationship study. J Neurol Sci 2014; 340: 99-102.
Wilson FC, Zangerle DJ, Darakjian AA, Bhutani M, Fliess JJ, Gehin JM, Strandes MW, Hamilton C, Sledge HJ, Hodge DO, Wang BW. Sex differences in self-reported symptoms and comorbidities associated with hypermobile Ehlers-Danlos syndrome and hypermobility spectrum disorders: A retrospective study. Research Square 2026; p. rs.3.rs-8310986.
Yoon HK, Lee HK, Jeon YT, Hwang JW, Lim SM, Park HP. Clinical significance of the cross-sectional area of the internal jugular vein. J Cardiothorac Vasc Anesth 2013; 27: 685-689.
Zhao S, Tran VH. Postural Orthostatic Tachycardia Syndrome. 2023 Aug 7. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2026. PMID: 31082118.
|







